33) Which law states that the specific heat of a gas remains constant at all temperatures and pressures
Reynault′s Law
Related Engineering Thermodynamics MCQ with Answers
both energy and mass cross the boundaries of the system
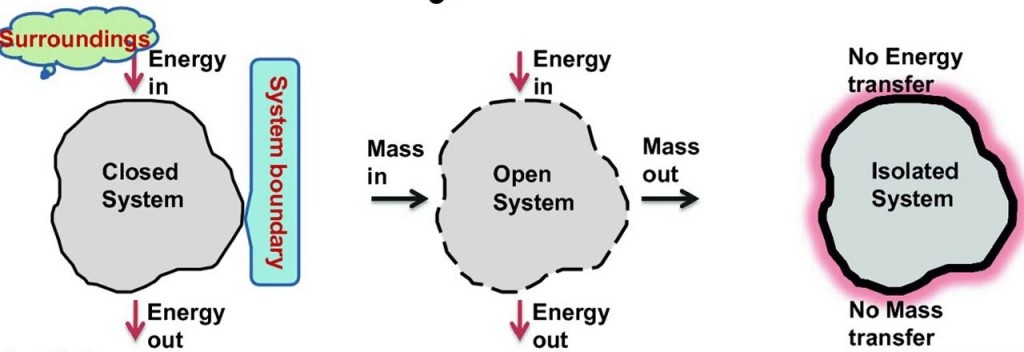
Classification of thermodynamic systems are
1. Closed system: Closed system is one in which boundary of the system does not allow the matter (mass) to cross it, then it is known as closed system. Here the system contains fixed or constant amount of matter (mass). Energy can cross the boundary. Heat and work are the only ways in which energy can be transferred between a closed system and its surroundings.
2. Open system: Open system is one in which boundary of the system allows the matter (mass) to flow into or out of the system, then it is known as open system. In open system matter (Mass) crosses the boundary of the system. Heat and work may also cross the boundary.
3. Isolated system: Isolated system is one in which boundary of the system does not allow the matter (mass) or the energy to flow into or out of the system, then it is known as isolated system. An isolated system in one which is completely uninfluenced by the surroundings. It is of fixed mass and no heat or work crosses the boundary of the system.
Charles′ law.
two values of specific heat
The specific heat of a substance my be broadly defined as the amount of heat required to raise the temperature of its unit mass through 1 degree. All the liquids and solids have one specific heat only. But a gas can have any number of specific heats depending upon the conditions, under which it is heated.
Specific heat at constant volume is the amount of heat required to raise the temperature of a unit mass of a gas throught 1 degree, when it is heat at constant volume.
Specific heat at constant pressure is the amount of heat required to raise the temperature of a unit mass of a gas through 1 degree, when it is heated at constant pressure.
The ratio of specific heat at constant pressure Cp and specific heat at constant volume Cv is always more than one.