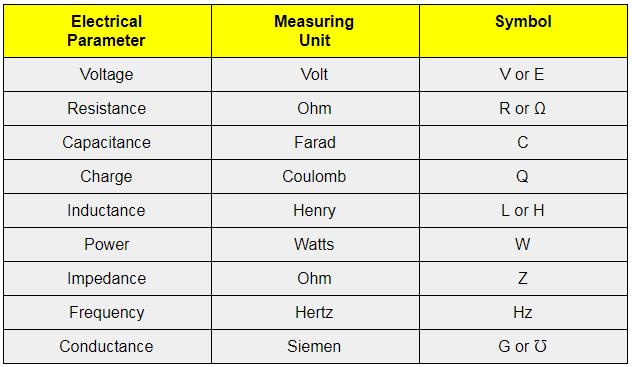
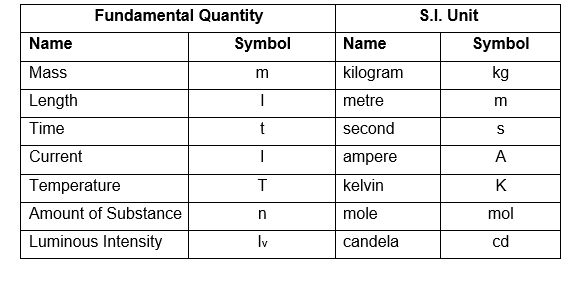
20) No liquid can exist as liquid at
zero pressure
Related Engineering Thermodynamics MCQ with Answers
a temperature of - 273.16°C
The lower the temperature, the lower the pressure inside of the sphere. If the exact values were plotted out, a linear relationship would be apparent. Extrapolating this line to the point where there would be no pressure yields absolute zero, which is about -273.15 degrees Celsius.
does not depend on the mass of the system, like temperature, pressure, etc.
Extensive properties The properties of the system, whose value for the entire system is equal to the sum of their values for the individual parts of the system, are called extensive properties.
For example, total volume, total mass and total energy of a system are extensive properties.
Intensive properties The properties of the system, whose value for the entire system is not equal to the sum of their values for the individual parts of the system, are called intensive properties.
For example, temperature, pressure and density of a system are intensive properties.